 Member has met all initial authorization criteria at the time of initial approval and.Note: The first two reauthorizations will each be for 6 months duration the third and all subsequent authorizations will be for 12 months duration.Ĭriteria for Continuation of Therapy at 6 months following initiation:Ĭontinued use of the requested medication at 6 months following initiation is considered medically necessary when all of the following criteria are met: Member must currently be enrolled in a randomized controlled trial conducted under an investigational new drug (IND) application or National Institutes of Health (NIH)-supported trial.Īetna considers all other indications as experimental and investigational.Īetna considers continuation of aducanumab-avwa (Aduhelm) therapy medically necessary for treatment of Alzheimer's disease (AD) when criteria are met.Member must have a recent brain magnetic resonance imaging (MRI) within one year prior to initiating treatment and.Have results from a lumbar puncture confirming the presence of elevated phosphorylated tau (P-tau) protein and/or elevated total tau (T-tau) protein, and reduced beta amyloid-42 (AB42) or a low AB42/AB40 ratio as determined by the lab assay detected in cerebrospinal fluid (CSF) and.Have a positron emission tomography (PET) scan confirming the presence of amyloid pathology or.Montreal Cognitive Assessment (MoCA) score of greater than or equal to 16 (see Appendix D) and.Mini-Mental Examination Status (MMSE) score of 21 - 30 (see Appendix C) or.Clinical Dementia Rating (CDR)-Global Score of 0.5 or 1 (see Appendix B) or.Member must have one of the following scores at baseline on any of the following assessment tools:.Member must have objective evidence of cognitive impairment at baseline (see Appendix A) and.Member must have mild cognitive impairment due to AD or mild AD dementia and.If less than 50 years of age, member has a genetic mutation in amyloid precursor protein ( APP), presenilin-1 ( PSEN1), or presenilin-2 ( PSEN2), or other clinical documentation to support early onset AD and.Member must meet one of the following criteria: This medication must be prescribed by or in consultation with a geriatrician, neurologist, psychiatrist, or neuropsychiatrist.Īetna considers aducanumab-avwa (Aduhelm) medically necessary for treatment of Alzheimer's disease (AD) when all of the following criteria are met: Aduhelm will not be used in combination with any other amyloid beta-directed antibodies (e.g., lecanemab).Bleeding disorder that is not under adequate control (including a platelet count less than 50,000 or international normalized ratio greater than 1.5).History of transient ischemic attacks (TIA), stroke, or seizures within the past 12 months.Requirement for therapeutic anticoagulation (e.g., anticoagulants, antiplatelets), except for aspirin at a prophylaxis dose or less (no more than 325mg daily).Suspected neurodegenerative etiology of cognitive impairment other than Alzheimer’s disease (AD), including but not limited to frontotemporal lobar degeneration (FTLD) or Lewy body disease (i.e., meeting consensus criteria for possible or probable dementia with Lewy bodies).For information on site of service for aducanumab-avwa (Aduhelm), see Utilization Management Policy on Site of Care for Specialty Drug Infusions.Ĭoverage will not be provided for members with any of the following conditions: Note: Site of Care Utilization Management Policy applies. For precertification of aducanumab-avwa (Aduhelm), call (866) 752-7021, or fax (866) 267-3277. For Statement of Medical Necessity (SMN) precertification forms, see Specialty Pharmacy Precertification. Precertification of aducanumab-avwa (Aduhelm) is required of all Aetna participating providers and members in applicable plan designs. 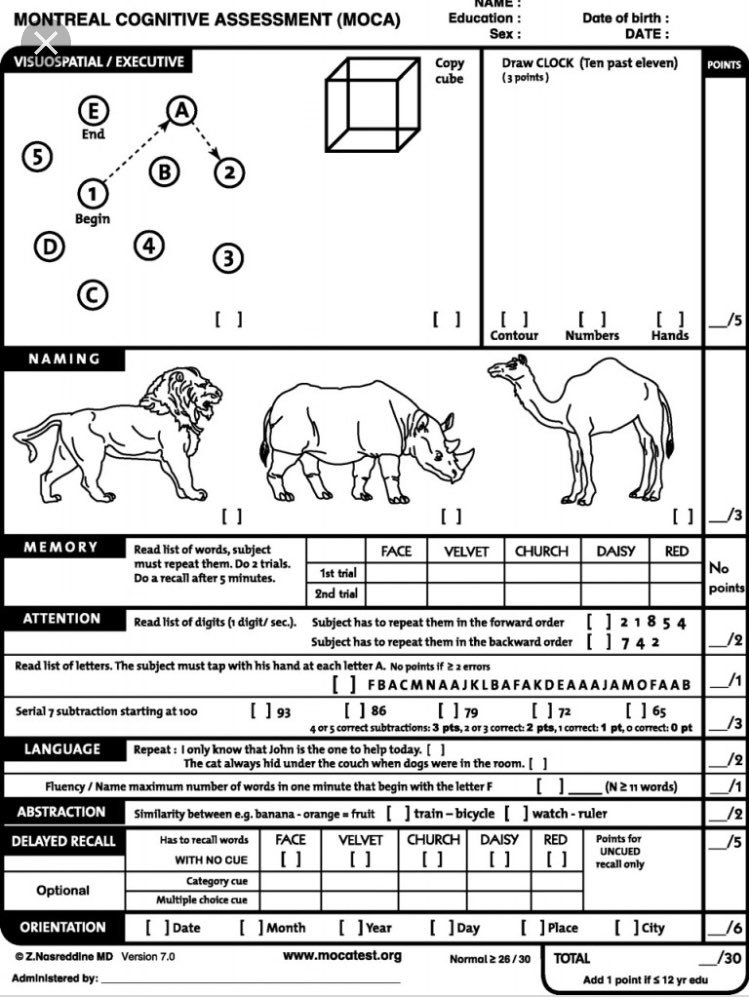
For Medicare criteria, see Medicare Part B Criteria. This Clinical Policy Bulletin addresses aducanumab-avwa (Aduhelm) for commercial medical plans. 
Number: 0996 Table Of Contents Policy Applicable CPT / HCPCS / ICD-10 Codes Background References 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
